APS Research & News
Peptides and Proteins
This article reflects work in the Raines Lab
Transforming growth factor-β (TGF-β) plays important roles in wound healing. The activity of TGF-β is initiated upon the binding of the growth factor to the extracellular domains of its receptors.
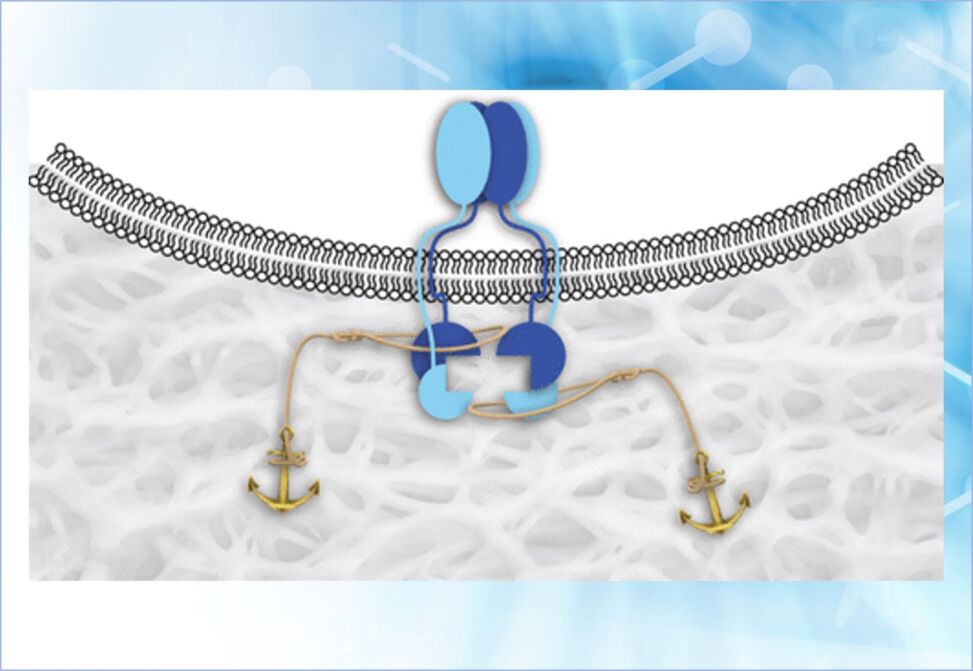
Chattopadhyay et al sought to facilitate the activation by clustering these extracellular domains. To do so, they used a known peptide that binds to TGF-β receptors without diminishing their affinity for TGF-β.
They conjugated this peptide to a collagen-mimetic peptide that can anneal to the damaged collagen in a wound bed. They find that the conjugate enhances collagen deposition and wound closure in mice in a manner consistent with the clustering of TGF-β receptors.
This strategy provides a means to upregulate the TGF-β signaling pathway without adding exogenous TGF-β and could inspire means to treat severe wounds.
Published here on March 5, 2022
Title: Bifunctional Peptide that Anneals to Damaged Collagen and Clusters TGF‑β Receptors Enhances Wound Healing
Authors: Sayani Chattopadhyay, Leandro B. C. Teixeira, Laura L. Kiessling, Jonathan F. McAnulty, and Ronald T. Raines*
Citation: ACS Chemical Biology 2022, 17, 2, 314-321